Newborn Screening
Newborn screening (NBS) is a vital public health program that enables the early detection of rare but serious conditions in infants, often before symptoms appear. For babies born with CCDS, early diagnosis through NBS can be life-changing. Timely intervention allows for the initiation of simple, cost-effective treatments that can prevent severe neurological damage, leading to improved health outcomes and quality of life.
What is Newborn Screening?
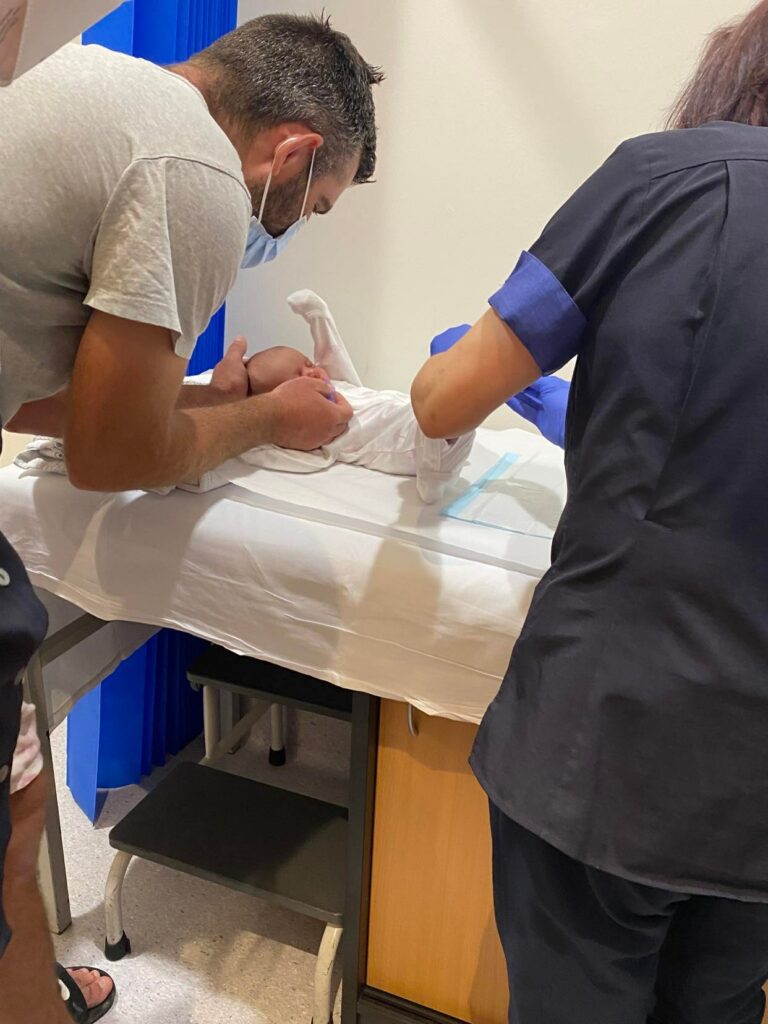
Newborn screening (NBS) is the collection of blood drops from a newborn’s heel onto a card of filter paper to screen for an approved group of disorders that require immediate treatment. The blood is usually collected between 24-48 hours after birth and sent to a government-run (or government-contracted) lab for testing.
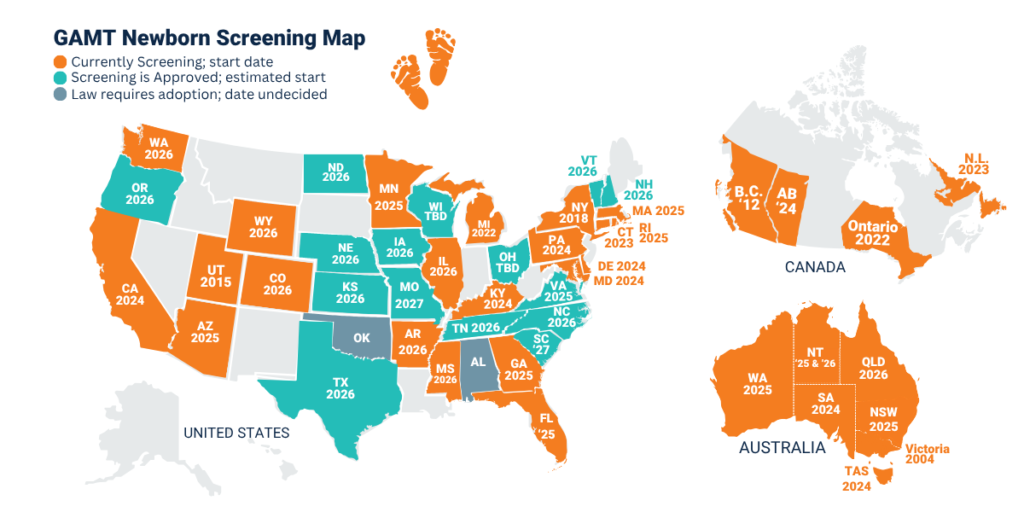
PKU is the most well-known disorder as it was the first disorder screened for, beginning in the 1960s, but most NBS panels now screen for many disorders. In the United States, the Secretary of Health and Human Services maintains a Recommended Uniform Screening Panel (RUSP) of disorders that is provided as guidance to the individual states. States are not required to follow the RUSP recommendations except in the case of those states that have passed legislation committing themselves to follow the RUSP. Inclusion on the RUSP is a major milestone for our community, achieved January 2023 and after nearly 7 years of advocacy work by the ACD and partners. RUSP inclusion increases the likelihood of GAMT being screened for, not only in the U.S., but in countries worldwide. This is evident in recent advancements seen on the map above.
ACD is actively advocating for the expansion of GAMT newborn screening and supports research of CTD newborn screening as well. AGAT newborn screening is theoretically possible with the measurement of GAA (also referred to as “GUAC” in newborn screening labs) and Creatine levels, the same levels measured in GAMT screening, but the sensitivity of the instruments currently available is not great enough to differentiate a “low-normal” and an AGAT level. There is hope that improvements in technology will make screening for AGAT possible.
Advocate with Us
If you’d like to advocate in your region, email us at info@creatineinfo.org and let’s work together! A local voice is the most influential in making change and we can help you prepare with slides and a customized presentation. Another way to advocate is to tell your story of GAMT and how it’s impacted you or a loved one’s life. Tag your stories on social media with #addGAMT or make a donation to support our work here.
Get Involved!
ACD supports the validation of testing for GAMT Deficiency and the research of plausibility for CTD and AGAT newborn screening by assisting interested labs in obtaining a very small portion of patients’ dried blood spots (DBS) that were collected shortly after birth. Many laboratories retain these DBS for years. If you’d like to help in this effort, please complete this form so that we can check if your child’s spot is available. If it is available, you will be contacted by ACD. You will decide if you wish to consent in the particular research being done. Filling out this form does not give us permission to act on your behalf. It does not allow us to use your child’s DBS without your consent. We will not share your information with anyone for any reason. The intent of this form is to identify the availability of DBS and allow you to decide what research you will support.
Additional NBS Info and Links
Publications & Materials
September 1, 2023
POSTER: Improved outcomes in early treated GAMT deficiency: A sibling study
ACD assisted Dr. Andreas Schulze in recruitment of sibling pairs for his study on the outcomes of early treated GAMT deficiency. He presented an informational poster on his findings at the Society for the Study of Inborn Errors of Metabolism (SSEIM) 2023 Annual Symposium in Jerusalem. This poster clearly shows the benefits of an early vs. late diagnosis of GAMT deficiency.
View the Poster
February 17, 2022
Modernization of Newborn Screening Study
ACD continues to contribute to newborn screening. Kim Tuminello, Founder and Director of Advocacy, participated in this study on modernizing newborn screening. See how a group of stakeholders discuss the impact of newborn screening at the federal and state levels, and the real possibility of 60 new gene therapies for rare diseases.
Read the Report
July 29, 2021
Prospective identification by neonatal screening of patients with guanidinoacetate methyltransferase deficiency
New York and Utah NBS programs share data on their screenings that ultimately resulted in the first and second diagnoses in the world of GAMT deficiency by newborn screening. ACD Executive Director, Heidi Wallis, is a co-author.
See the abstract on PubMed.
